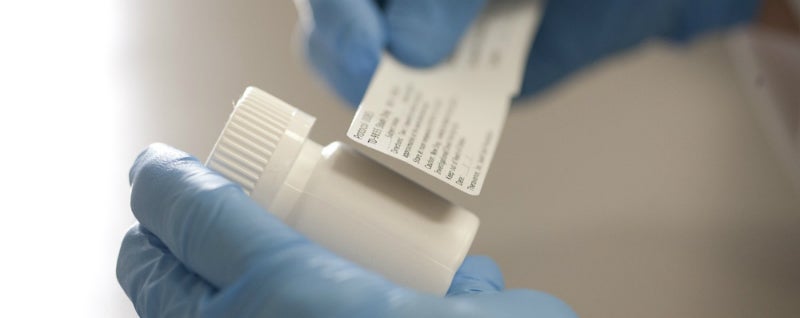
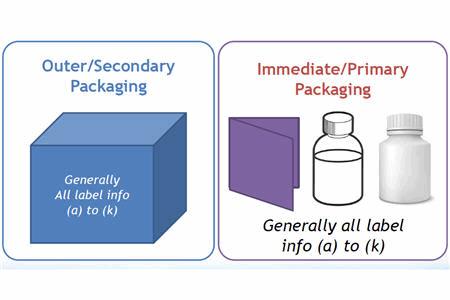
Investigational Drugs: Strategies for Sponsors, FDA, and Clinical Sites to Prevent Product-Related Errors (Part II) | Institute For Safe Medication Practices

Study of Chinese mRNA vaccine candidate shows booster raises effectiveness against Omicron | South China Morning Post

Solve Expiry Labels, DtP, and Timelines for EU 536/2014 Clinical Trials Regulation | Healthcare Packaging