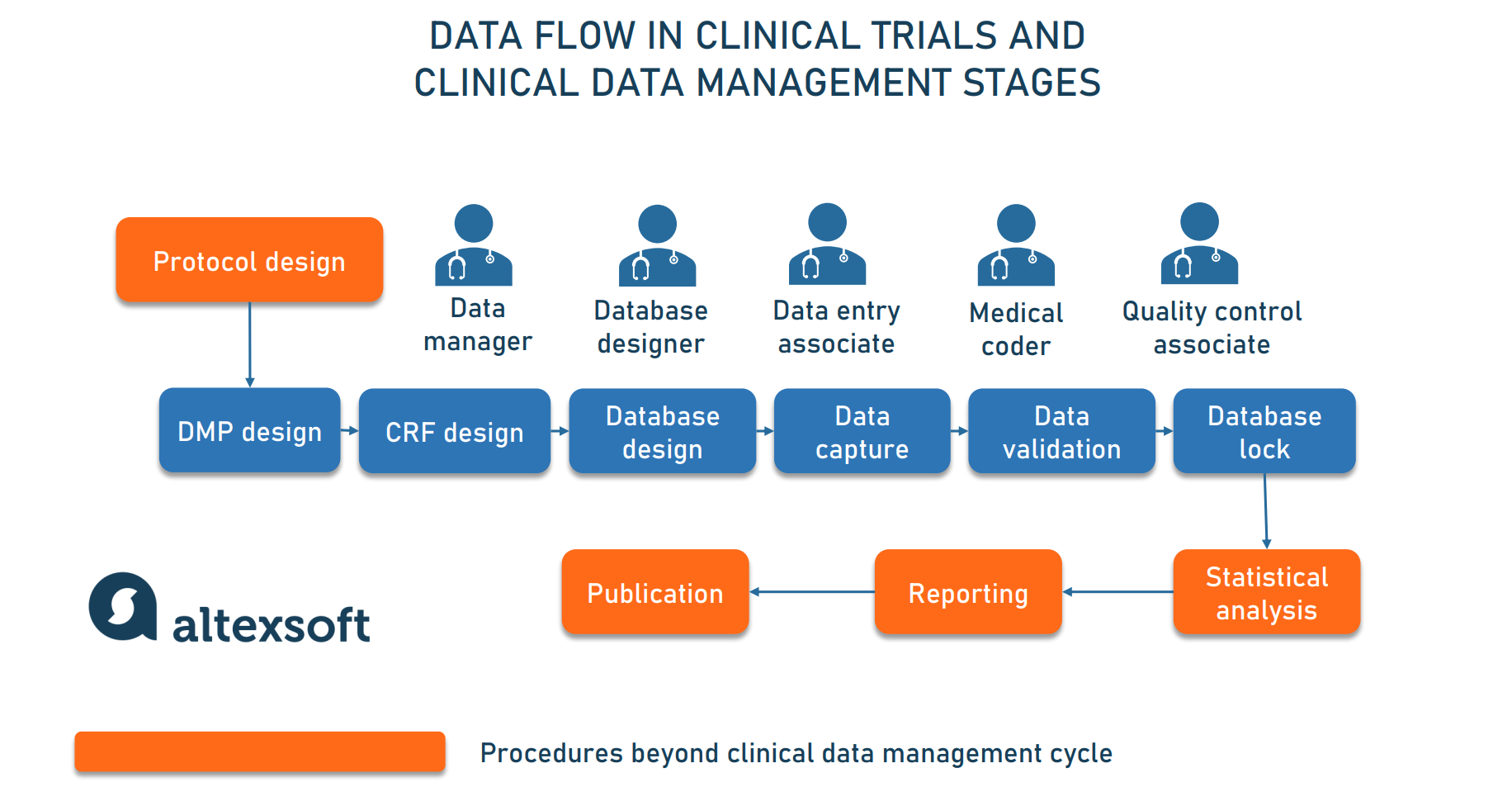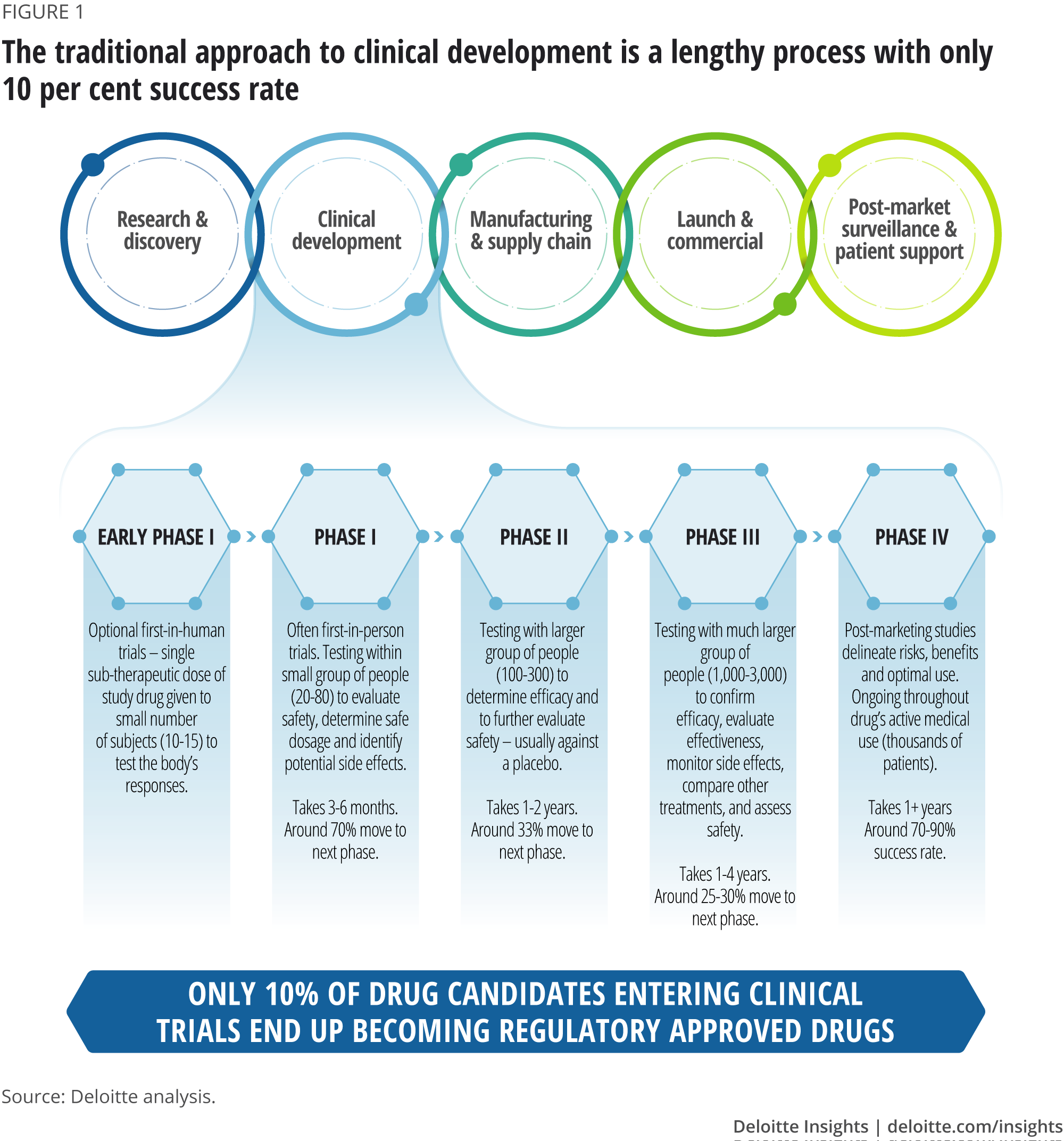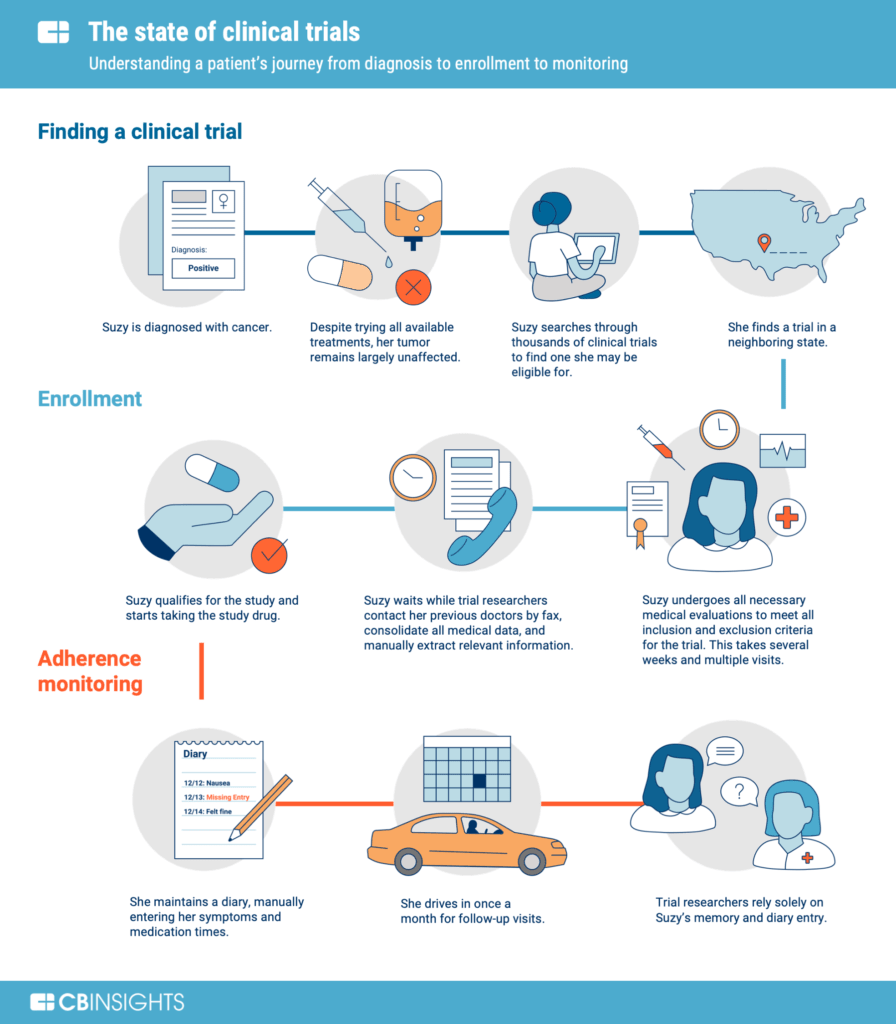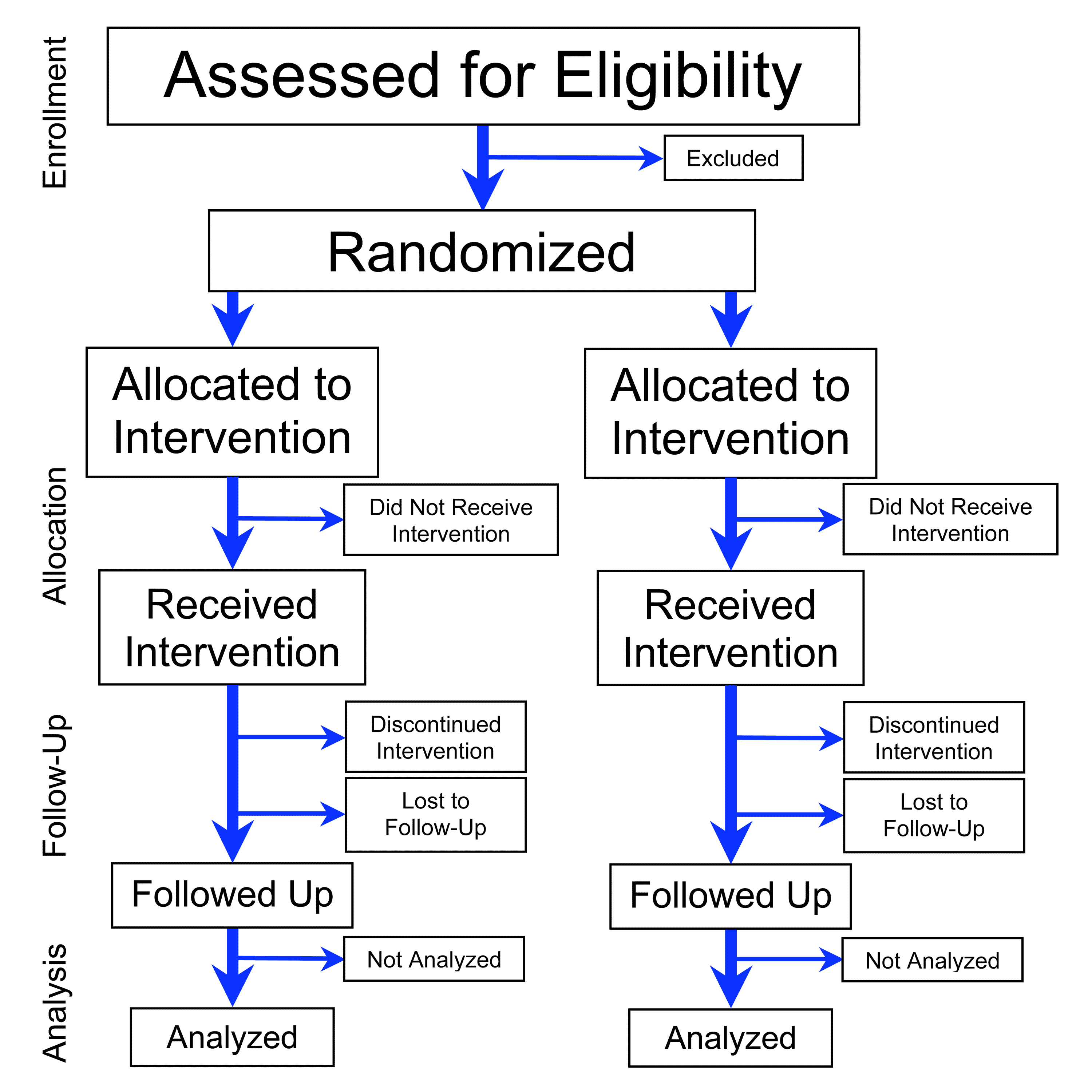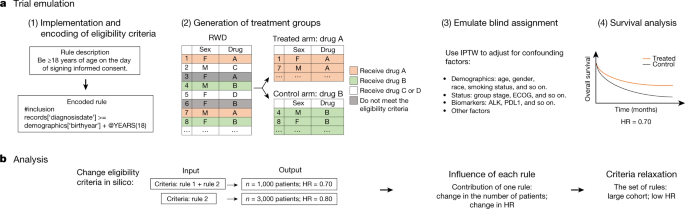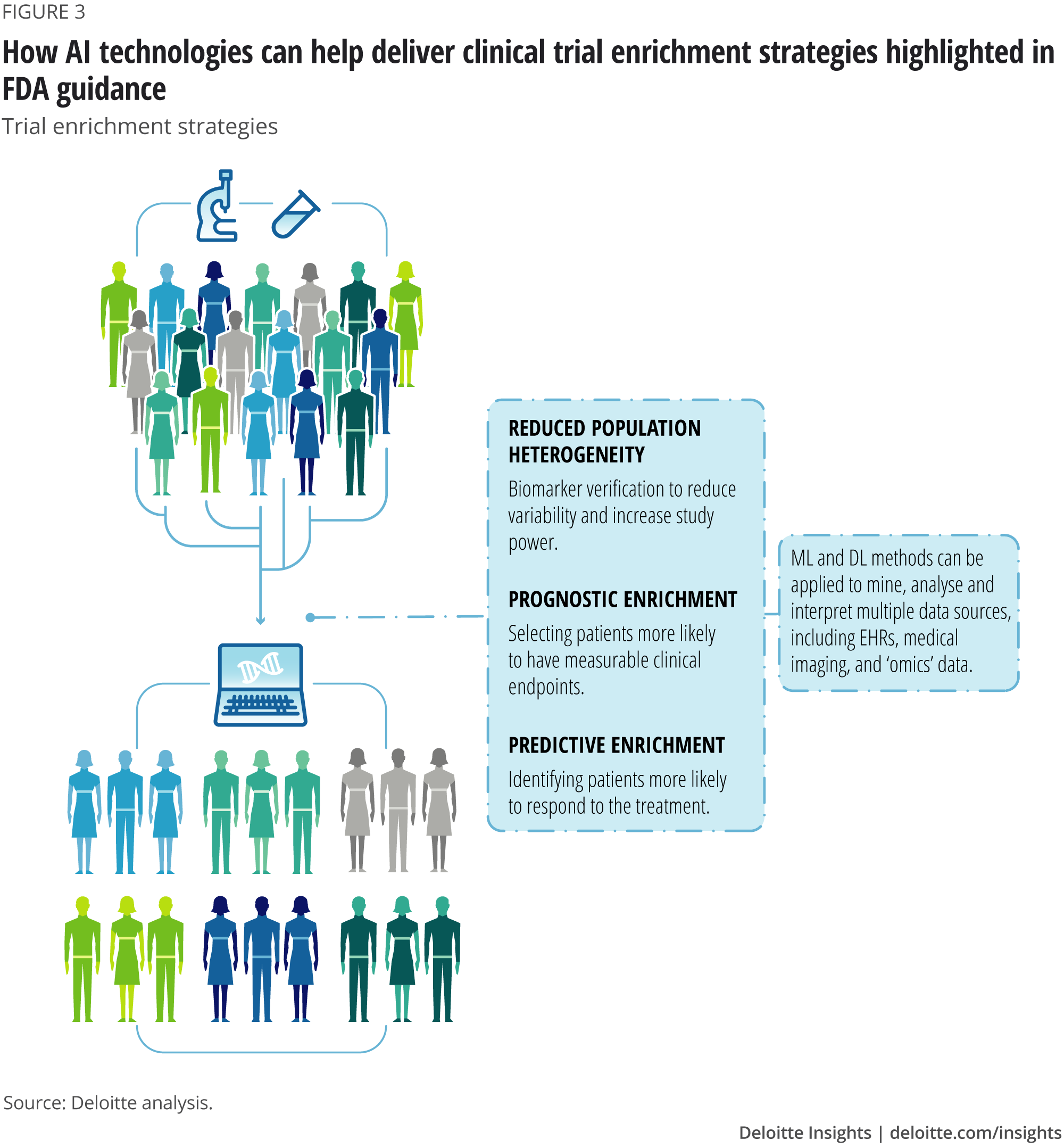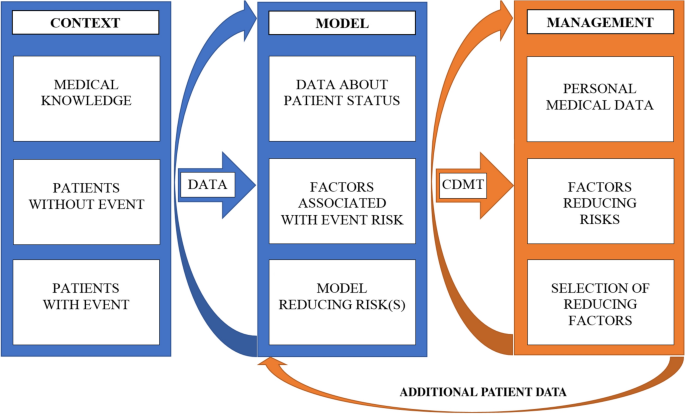
A theoretical model of health management using data-driven decision-making: the future of precision medicine and health | Journal of Translational Medicine | Full Text

2017 Cardiovascular and Stroke Endpoint Definitions for Clinical Trials | Journal of the American College of Cardiology

Schedule of Events. Example visit and assessment specification from a... | Download Scientific Diagram

Understanding pre-exposure prophylaxis (PrEP) regimen use: Switching and discontinuing daily and event-driven PrEP among men who have sex with men - eClinicalMedicine

Aligning Trial Design and Key Processes in Phase III Event Driven Trials: Protocol (via a Special Protocol Assessment), Data Monitoring Committee Charter. - ppt download

Rationale and design of the Pemafibrate to Reduce Cardiovascular Outcomes by Reducing Triglycerides in Patients with Diabetes (PROMINENT) study - ScienceDirect
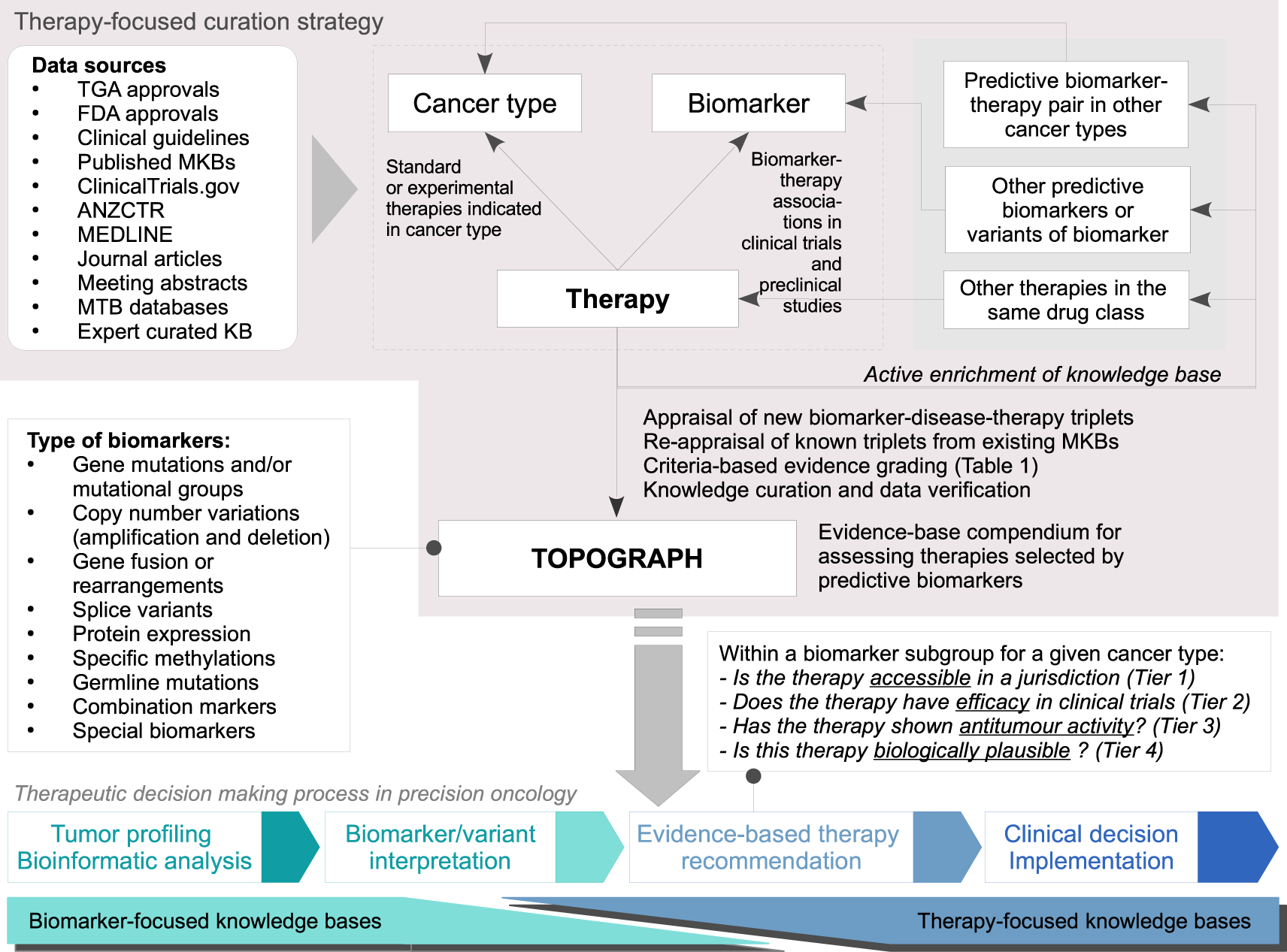
Criteria-based curation of a therapy-focused compendium to support treatment recommendations in precision oncology | npj Precision Oncology

Classical vs. event-driven, GSD design of a hypothetical clinical trial... | Download Scientific Diagram